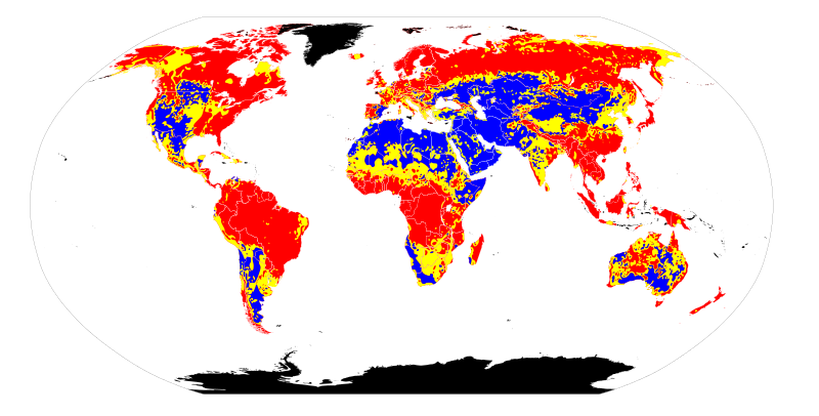
 Soil pH maps from World. Author: Ninjatacoshell Soil pH maps from World. Author: Ninjatacoshell Soil acidification is the buildup of hydrogen cations, also called protons, reducing the soil pH. This happens when a proton donor gets added to the soil. The donor can be an acid, such as nitric acid and sulfuric acid (these acids are common components of acid rain). It can also be a compound such as aluminium sulfate, which reacts in the soil to release protons. Many nitrogen compounds, which are added as fertilizer, also acidify soil over the long term because they produce nitrous and nitric acid when oxidized in the process of nitrification. Acidification also occurs when base cations such as calcium, magnesium, potassium and sodium are leached from the soil. This leaching increases with increasing precipitation. Acid rain accelerates the leaching of bases. Plants take bases from the soil as they grow, donating a proton in exchange for each base cation. Where plant material is removed, as when a forest is logged or crops are harvested, the bases they have taken up are permanently lost from the soil. Pollution Acidification may also occur from nitrogen emissions into the air, as the nitrogen may end up deposited into the soil. 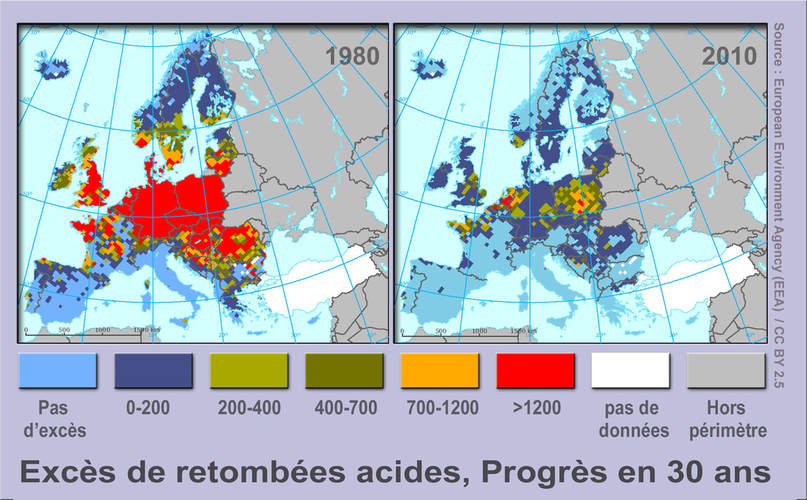 AcidificationExcèsUE AEEfr.jpg / Auteur: Lamiot AcidificationExcèsUE AEEfr.jpg / Auteur: Lamiot In Europe, the reduction of input and the effort of acid deposition (oil, gas désoufrés, descending coal) is successful (for SO2 and some volatile organic compounds, including at least NOX (due to transport and fertilizer), and NH 3. Overtaking "load crititique acid settling" is decreasing (unit: equivalent ha -1 y -1). The European Environment Agency's attention on the map, for the value of the 2010 target value by insisting on providing the implementation of the NEC Directive (the target may not have arrived) Soil Conditioner is a new solution for treating - LINK SOILIt is known that about 10% of the agricultural area is suffering from saline-alkaline soils and the area affected by saline-alkaline conditions is growing by approximately 14% every year in the world. Usually two methods are mainly used to treat saline-alkaline soils at the moment.
Irrigation (washing the soil with water) the disadvantages of this method are huge quantities of water are needed and by using this method you transfer the salinity problem to other areas. Chemical treatment - big disadvantages as well: - very expensive - create problems to environment - reducing and eliminating micro-organisms in Soil. Now a new method has been developed by our scientists. LINK-SOIL is a biological-based product, using complex agents in an organic-biochemical formulation that act with the salt ions in the soil, binding the salt into other (bigger) molecules. With water, LINK-SOIL moves the salinity deeper, removing the harmful salt content from roots area. These will then help to: • enhance soil micro environment • provide good soil moisture • create root penetration • provide water infiltration and aeration • enhance higher seeds germination • resulting in healthy growth and increasing yield At the same time iron (Fe), manganese (Mg), phosphorus (P) and other nutrition elements are added to the soil. The same good effect applies for alkali soil. Comments are closed.
|
- HOME
- COMPANY
-
Showroom
- TAIWAN TEA >
-
KITCHEN UTENSILS
>
- Tornado Potato Twister Cutter JF-9901
- Japanese Turning Vegetable Slicer YT-8602
- Turning Type Vegetable Shredder with Cordless Screw Driver ST-9705
- Vegetable Mandolin Slicer YT-9104
- Super Mandolin Slicer (Jumbo) ST-9804
- Vegetable Peeler Blades (Serrated & Julienne) ST-9701
- Serrated & Julienne Peelers ST-9308
- BATHROOM EQUIPMENT >
- NATURE FERTILIZERS >
- NATURAL PESTICIDES >
- SOIL CONDITIONER >
- OTHER >
- DOWNLOAD
- Contact us
- NEWS / EVENTS
- Q & A
|

 RSS Feed
RSS Feed


